Create Jira Ticket
Something went wrong.
This idea cannot be sent to Jira.
This idea cannot be sent to Jira.
Intelligent capture and routing of data from any edge diagnostic devices/test kits
With TIBCO platform, you can register and interact with IoT Device Farm to process derived data and store the data as needed to analyze and address a large number of analytical use cases extending any layer of its architecture.
Specifically, TIBCO offers open source, cloud-native, and vendor neutral platform with the following value-add capabilities:
- Automate data capture directly from IVD devices
- Standardize data elements, formats, and payloads
- Receive data from any device, located anywhere
- Minimize manual data entry
- Common entry point for all reporting systems (IVDs, apps, etc)
- Centralized access and management of all edge devices
- Collaborate with LF Edge and other relevant open source projects to ensure consistency and interoperability across the IoT
Please see below URL on a quick demonstration on integrating any edge device for data collection and processing:
For solution page and GitHub site, please access the following URL:
edited on Nov 17, 2020 by
Jimmy Cesario

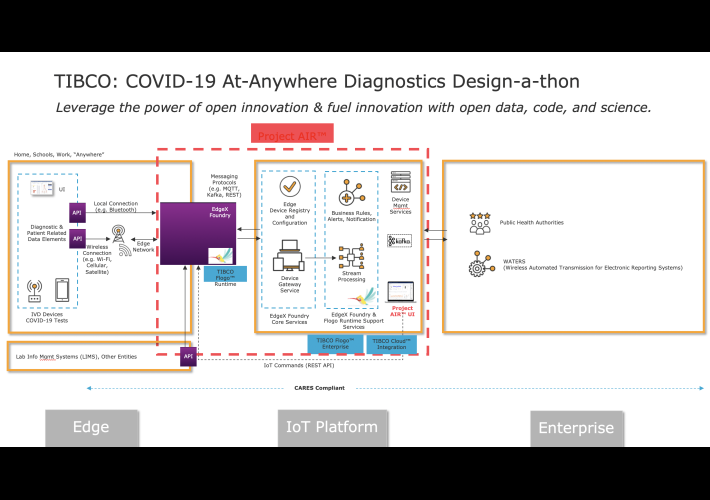


Project Air Reference Architecture
Have you considered a FHIR API or some sort of bridge software?
Thanks for the videos! Nice approach as it seems to meet a number of the requirements.
Are there limits to the types/kinds of devices? Can it support different smartphones that might serve as IVD like in "interpreting" patient performed results? How about different lab/IVD Point of Care Testing devices/kits? (Interested in how CLIA compliant lab reporting would be supported too.)
Seems like the products need to be installed at a health system/contracted entity? Would it work for individuals not associated with health system (i.e. patient performed testing at home)? Scalable nationally or limited to contracted/paid customers?
Looks like the vendor agnostic approach would support a variety of health professional/lab IVD vendor devices performed in lab or point of care testing, as well as patient performed kits/smartphone readers/apps, as well as direct data transfer from fitbit, finger pulse oximeters (oxygen levels) or other wearable data such as those devices that have detected asymptomatic/minor symptomatic cases in PGA players.